Why Power Supply Design Is Critical for Medical Device Safety and Performance
No matter how sophisticated an electronic device may be, it’s ultimately useless without a stable and reliable power source. Power supplies are essential for converter energy from the grid or a battery into a regulated, usable voltage for electronic components, but they also serve as a barrier between potentially dangerous power spikes, making them critical for sensitive or safety-critical systems.
As a result of this dual role, power supplies are almost always subject to safety standards, especially those involved with medical devices where devices operate in close proximity to patients (often in direct contact), and failure can result in injury, misdiagnosis, or worse.
So, what exactly sets medical power supplies apart from those used in other industries? And what are the five key considerations engineers must address when designing or selecting a power supply for medical use?
Understanding IEC 60601-1 and IEC 60601-1-11
When it comes to power supplies used in medical devices, two international standards stand out as the foundation of safety compliance: IEC 60601-1 and IEC 60601-1-11.
IEC 60601-1 is the most foundational standard, and outlines the general requirements for basic safety and essential performance in medical electrical equipment. It covers everything from insulation strength and creepage distances to leakage current limits and protective earth requirements. The primary goal of IEC 60601-1 is to ensure that medical devices are designed and built in a way that prevents electrical shock, fire, and mechanical hazards under both normal and single-fault conditions. Examples of devices that fall under this scope include ECG machines, defibrillators, and blood pressure monitors.
IEC 60601-1-11, however, extends these requirements to medical equipment intended for use in home healthcare environments (i.e. where patients will be responsible for operating their own equipment). Unlike hospitals or clinical settings, home environments present an uncontrolled environment, where fluctuating power quality, limited operator training, and potential exposure to moisture, dust, or mechanical stress exist. Examples of devices covered by IEC 60601-1-11 include portable oxygen concentrators, dialysis systems, and infusion pumps.
| Standard | Scope | Environment | Example Devices |
|---|---|---|---|
| IEC 60601-1 | General safety and performance | Clinical settings | Dialysis machines, ECG machines, defibrillators, blood pressure monitors |
| IEC 60601-1-11 | Home-use medical equipment | Uncontrolled homes | Oxygen concentrators, dialysis systems, baby moniters |
Table 1. Comparison of IEC 60601-1 and IEC 60601-1-11 standards for medical device safety.
Why These Standards Matter for Medical Power Supplies
Whether a power supply is delivering stable voltage to a life-support machine or charging a portable diagnostic device, the performance of such supplies directly impacts patient safety and clinical reliability. However, even if a power supply is built to high standards or manufactured by a reputable supplier, that doesn’t automatically qualify that supply for medical environments.
Engineers selecting supplies for medical environments must look far beyond datasheet specifications, making sure to verify safety certifications, insulation ratings, and leakage current limits. Power supplies in these applications need to also be able to handle the practical realities of medical environments, including fluctuating input voltages, exposure to cleaning agents, and potential interference from other household or hospital devices on the same power grid.
Top Five Power Supply Considerations for Medical Applications
Number One – Isolation and Means of Protection (MOPP)
The most important factor in medical power supply design is isolation; the physical and electrical separation that prevents dangerous voltages from reaching the patient or operator. Any failures or faults in a power supply must never propagate into the medical device circuitry or the person using it.
IEC 60601-1 defines isolation requirements through the concept of Means of Protection (MOP), which can apply to both patients (MOPP) and operators (MOOP). Depending on the application, a supply may require one or two Means of Patient Protection, which correspond to specific insulation grades, creepage distances, and dielectric strength levels.
To meet these standards, medical-grade power supplies must incorporate:
- Reinforced or double insulation to prevent voltage breakdown across barriers
- High-isolation transformers that maintain electrical separation between input and output
- Proper grounding paths to safely redirect fault currents away from the user
- Fault containment systems that prevent internal failures from propagating to the device or patient
These design elements work together to ensure that even under single-fault conditions, the power supply remains safe and compliant.
Number Two – Reliability, Redundancy, and Protection Features
Medical environments represent one of the highest-risk use cases for electronic systems, where a single malfunction can interrupt a life-sustaining process, distort a critical measurement, or disable equipment at the worst possible moment. In these situations, the power supply becomes critical to system reliability, and its failure is never an option.
To meet such demands, medical power supplies need to be designed with substantial safety margins and exceptional long-term durability. Higher Mean Time Between Failures (MTBF) figures, highly conservative component derating, and the use of premium-quality components including capacitors, transformers, and semiconductors are all essential for ensuring continuous operation.
Comprehensive protection circuitry is also critical, and such supplies must be able to defend against overvoltage, overcurrent, short circuits, and thermal overload, all while providing graceful recovery. Additionally, electrostatic discharge (ESD) and brownout protection help guard against unpredictable real-world conditions such as static buildup or unstable mains power.
Number Three – Leakage Current and Applied Part Classification
Leakage current (unwanted current that flows through insulation or grounding paths) is a serious concern in medical electronics. Even tiny amounts of leakage current can be dangerous, especially in systems connected directly to a patient’s body (for example, ECGs create conductive pathways across the heart, which can be stopped with a few milliamps of current).
To manage this risk, medical devices are classified into Type B, BF, and CF categories, based on how and where they contact the patient. Type B devices are general body-contact systems, Type BF are floating body-contact systems, and Type CF, the most stringent, are cardiac floating systems requiring the lowest allowable leakage currents.
| Classification | Patient Contact Type | Leakage Current Limit | Example Devices |
|---|---|---|---|
| Type B | General body contact | Highest | Ultrasound machines |
| Type BF | Floating body contact | Moderate | Blood pressure monitors |
| Type CF | Cardiac contact | Lowest | ECG machines |
Table 2. Classification of medical devices based on patient contact type and allowable leakage current.
Power supplies, therefore, must be designed and certified to meet such limits. In these scenarios, the distinction between Class I (earth-grounded) and Class II (double-insulated) power supplies also plays a serious role in determining how fault currents are managed and isolated from the patient or operator.
Number Four – EMC Immunity
Electromagnetic compatibility (EMC) is critical in most electronic circuits, but when dealing with life-saving equipment, it becomes even more paramount. The modern healthcare environment is saturated with potential sources of interference, including Wi-Fi routers, Bluetooth peripherals, mobile phones, computers, and wireless telemetry systems. Every one of these devices can disrupt sensitive medical equipment if the attached power supply isn’t properly shielded or filtered.
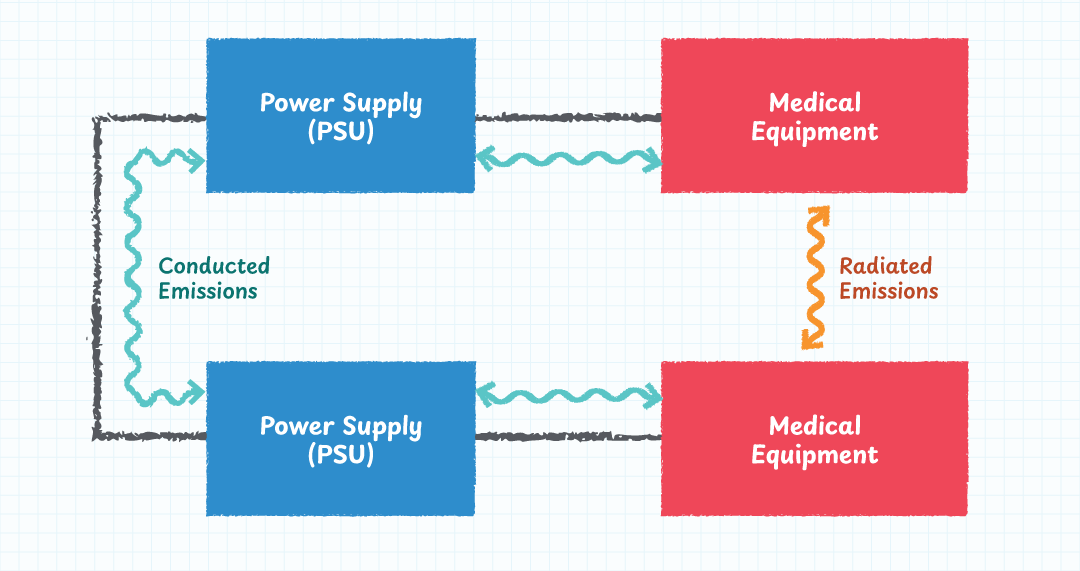
A medical-grade power supply must therefore exhibit significant immunity to external interference while also maintaining low emissions so as to minimise disturbance to other nearby medical devices. Achieving this balance requires robust EMI filtering, careful PCB layout, proper grounding, and compliance with the relevant EMC standards for medical equipment.
Number Five – Environmental Considerations
A power supply’s performance is always influenced by its environment, and in medical applications, temperature, humidity, altitude, and air pressure can all affect safety. A supply designed for a controlled hospital setting may behave very differently when used in a home environment, especially when exposed to poor ventilation, fluctuating mains voltage, or household contaminants such as dust and cleaning agents.
In home healthcare settings, where environments are less controlled than hospitals, designers need to account for several critical factors:
- Tolerance to environmental extremes such as temperature, humidity, altitude, and air pressure is essential for safe operation.
- Filtering for household electrical noise protects against voltage spikes and transients from appliances like microwaves or washing machines.
- Insulated enclosures ensure touch safety, preventing accidental contact with hazardous voltages.
- User-friendly design is critical, as many devices are operated by non-professionals including elderly patients, children, or caregivers.
Considering that medical devices often need to be physically handled, touch safety is another critical factor. Medical devices used at home are often operated by non-professionals, including the elderly, children, or caregivers, meaning that enclosures must be fully insulated and designed to prevent contact with hazardous voltages under any circumstance.
Designing Medical Power Supplies for Safety, Compliance, and Real-World Resilience
Power supplies used in the medical industry need to meet extremely high standards to ensure safety and reliability. Furthermore, they also need to be exceptionally robust, able to withstand environmental stress, electrical interference, and operational misuse.
By adhering to the stringent requirements of IEC 60601-1 and IEC 60601-1-11, and by sourcing components from reputable, medically certified manufacturers, designers can help ensure that medical equipment performs safely and reliably throughout its operational life.

.png)





